The pandemic has brought about a revolution in the field of research, especially in pharmacology. Research studies aimed at finding an effective solution against COVID-19 have increased exponentially since the start of the pandemic. Various vaccines are now on sale and have themselves become the subject of numerous research studies. These studies seek to monitor the safety, efficacy, stability, and other aspects of these drugs.
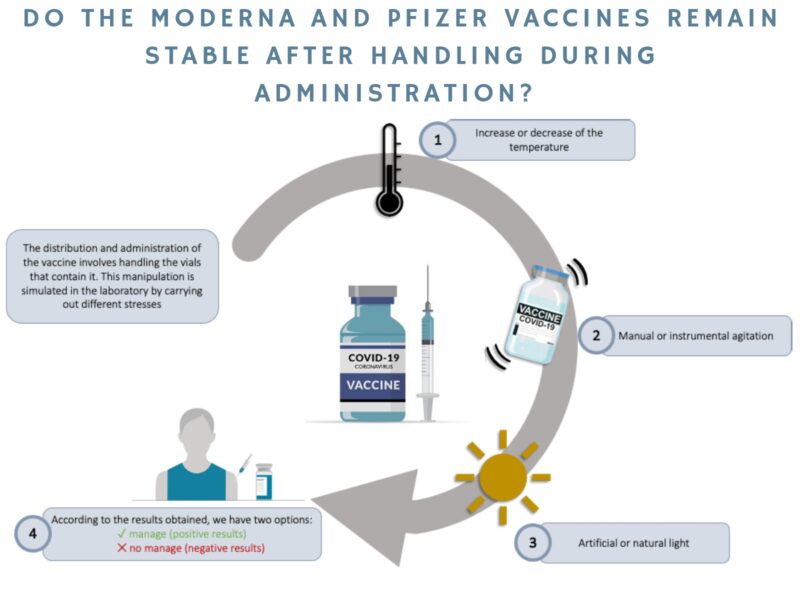
This research studies the stability of the Moderna and Pfizer vaccines during distribution and handling in hospital settings to assess whether they maintain their physicochemical properties. To this end, various different stress tests are carried out on the vaccines in the laboratory. These are similar to the stresses to which the vaccines may be exposed during distribution and handling. They are also subjected to more violent stress tests, such as shaking, high temperatures, and exposure to natural or artificial light, among others so as to verify that the particulate that makes up the vaccine remains stable. The aim of this research is to find out whether we can reduce the strict measures that must be followed when distributing the vaccines and administering them to the population.
Keywords: stability; physicochemical properties; vaccines; COVID-19
Directed by: Natalia África Navas Iglesias
